In this article, I will discuss the oxidation of alcohols. Here, we will look at how primary, secondary, and tertiary alcohols behave differently during oxidation reactions.
Oxidation and α-Hydrogens.
When alcohols are treated with oxidizing agents different types of alcohols react differently because of the different number of α-hydrogens (hydrogens attached to the carbon atom bearing the hydroxyl group) present in each type of alcohols.
Oxidation of Primary Alcohols.
A primary alcohol which has two α-hydrogens, is oxidized to an aldehyde. The aldehyde thus formed still has an α-hydrogen and can be further oxidized to a carboxylic acid. The oxidation of aldehydes to carboxylic acids usually requires milder oxidizing agents than those required for the oxidation of primary alcohols to aldehydes. The oxidation of primary alcohols therefore does not stop at the aldehyde stage, but proceeds on to the formation of carboxylic acids.
\underset{\text{A primary alcohol}}{\mathrm{RCH_2OH}}
\xrightarrow{[O]}
\underset{\text{An aldehyde}}{\mathrm{RCHO}}
\xrightarrow{[O]}
\underset{\text{A carboxylic acid}}{\mathrm{RCOOH}}
However, an advantage can be taken of the fact that the aldehydes generally have lower boiling points than the corresponding alcohols (why ?), and can be removed from the reaction mixture by distillation as soon as they are formed. This is applicable only to those aldehydes which boil significantly below 100°C. In other cases, this method is used for the oxidation of primary alcohols to carboxylic acids.
Oxidation of Secondary Alcohols.
The secondary alcohols which have only one α-hydrogen are oxidized to ketones and the reaction usually stops at this stage.
\underset{\text{A secondary alcohol}}{
\mathrm{
\begin{array}{c}
R \\
| \\
CHOH \\
| \\
R
\end{array}
}}
\xrightarrow{[O]}
\underset{\text{A ketone}}{
\mathrm{
\begin{array}{c}
R \\
| \\
C{=}O \\
| \\
R
\end{array}
}}
Oxidation of Tertiary Alcohols.
Tertiary alcohols have no α-hydrogen, and therefore are generally resistant to oxidation. However, on prolonged heating with acidic oxidizing agents, the tertiary alcohols are dehydrated to alkenes which undergo oxidative degradation through cleavage at the carbon-carbon double bond, to yield carbonyl compounds of lower molecular weights.
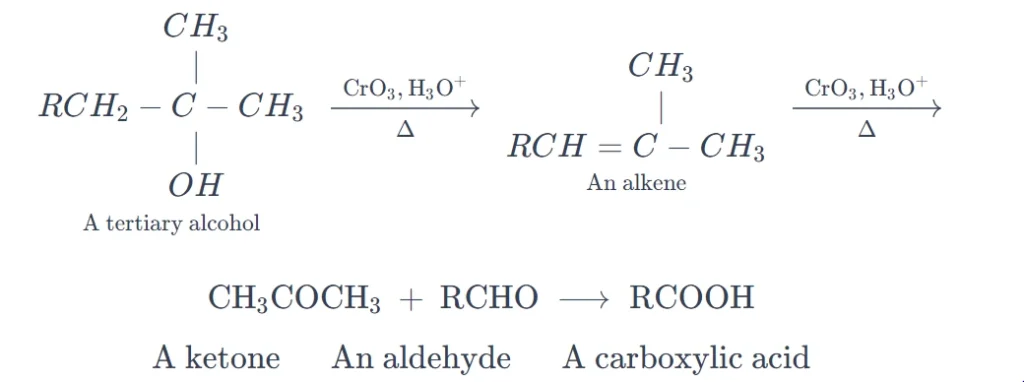
Common Oxidizing Agents.
Oxidation of primary and secondary alcohols in the laboratory can be carried out by various oxidizing agents, but the most commonly used oxidizing agent is chromic acid, H2CrO4, which is generally formed when chromic oxide, CrO3, or potassium dichromate, K2Cr2O7, is added to sulfuric acid, or chromic oxide to glacial acetic acid. The chromic acid oxidation is particularly useful for secondary alcohols due to the stability of the product, ketones, to these conditions. The reaction proceeds through an initially formed chromate ester.
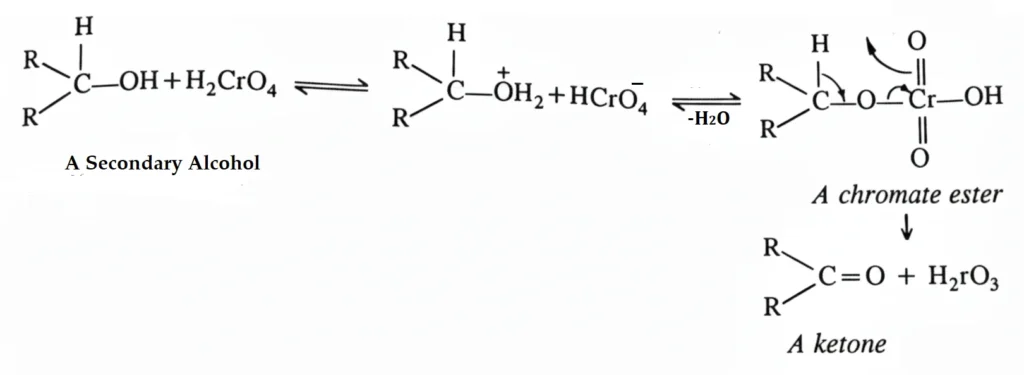
Oxidation of secondary alcohols are generally carried out in acetone or acetic acid solutions, acetic acid being the most generally used solvent for such reactions. Chromic oxide and sulfuric acid in aq. acetone is called the Jones reagent. The Jones reagent has no effect on the double bonds present in the molecules.
Pyridinium Chlorochromate (PCC) – A Selective Oxidizing Agent.
An excellent reagent for the oxidation of primary alcohols to aldehydes in the laboratory is pyridinium chlorochromate (PCC) which is formed when chromic oxide is dissolved in HCl and then treated with pyridine.
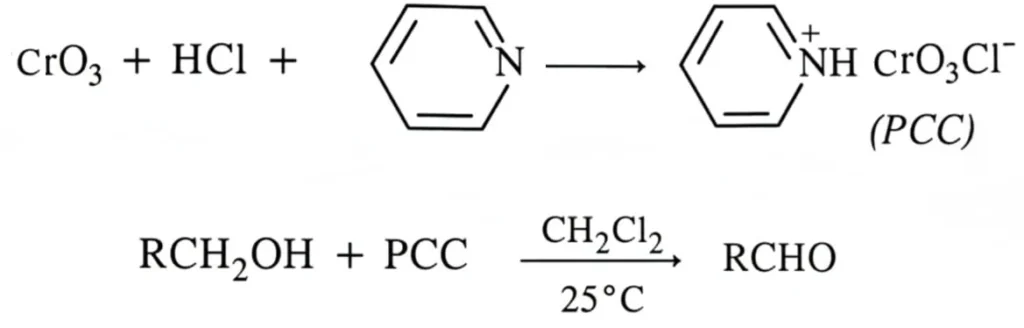
PCC also does not affect the double bonds.
Oxidation Using Potassium Permanganate (KMnO₄).
Oxidation of primary alcohols to carboxylic acids is generally carried out by alkaline aqueous potassium permanganate. Manganese dioxide, MnO2, produced during this reaction precipitates out under basic conditions, and can be removed by filtration after the oxidation is complete. The filtrate on acidification yields the carboxylic acid.
Neutral KMnO4 oxidizes unsaturated groups more rapidly than alcohols.
Catalytic Dehydrogenation of Alcohols.
Primary and secondary alcohols can also be converted to aldehydes and ketones, respectively, on industrial scale, by catalytic dehydrogenation. (Dehydrogenation of organic compounds corresponds to oxidation, and hydrogenation to reduction). Dehydrogenation of alcohols is most conveniently affected by passing the vapours of the alcohols over a catalyst heated to about 300°C. A very effective catalyst consists of oxides of copper and chromium, CuO.Cr2O3, and is known as Adkin’s catalyst.
\text{RCH}_2\text{OH} \xrightarrow[\text{300}^\circ\text{C}]{\text{CuO}\cdot\text{Cr}_2\text{O}_3} \text{RCHO} + \text{H}_2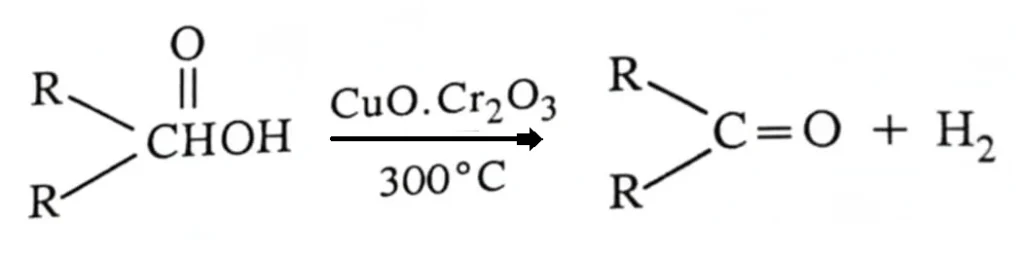
Methanol gives formaldehyde which under the conditions of the reaction decomposes to carbon monoxide and hydrogen. However, formaldehyde can be obtained by passing a mixture of methanol and air over a suitable catalyst at an elevated temperature where hydrogen is converted to water.
\text{CH}_3\text{OH} + \frac{1}{2}\text{O}_2 \xrightarrow[\text{300}^\circ\text{C}]{\text{Catalyst}} \text{HCHO} + \text{H}_2\text{O}